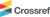Expression of Cyclooxygenase-2 by 4-Hydroxynonenal in Vascular Smooth Muscle Cells
Abstract
This study aimed to investigate the mechanism by which 4-hydroxynonenal (HNE), a key lipid peroxidation product, promotes vascular dysfunction, including that observed in diabetic retinopathy, which is a leading cause of blindness in the working-age population. The focus was specifically on the effect of HNE on cyclooxygenase-2 (COX-2) expression and nitric oxide (NO) generation within vascular smooth muscle cells (VSMCs).
VSMCs were treated with HNE, and the effects were analyzed using western blotting for protein expression, reverse transcriptase-polymerase chain reaction (RT-PCR) for mRNA levels, cellular fractionation for extracellular signal-regulated kinase (ERK) localization, and specific inhibitors (U0126, SB203580, and indomethacin) to delineate the signaling pathways. Intracellular NO formation was measured using flow cytometry with 4,5-diaminofluorescein (DAF-2) as a specific NO indicator.
HNE treatment significantly increased COX-2 expression and NO production in VSMCs. This effect was mediated through the ERK pathway, as evidenced by HNE-induced ERK phosphorylation, nuclear translocation, and complete inhibition of both COX-2 and NO generation by the ERK inhibitor U0126. The p38 MAPK inhibitor SB203580 and the COX-2 inhibitor indomethacin did not suppress HNE-induced NO production, indicating that ERK activates COX-2 and NO through independent pathways.
This study demonstrated that HNE induces COX-2 expression and NO production in VSMCs via the ERK signaling pathway, suggesting that targeting the upstream HNE-ERK signaling axis could be a promising therapeutic strategy for diabetic vascular complications.
초록
본 연구는 지질 과산화의 주요 생성물인 4-hydroxyonenal(HNE)가, 근로 연령층에서 실명의 주요 원인이 되는 당뇨망막병증에서 관찰되는 혈관 기능장애를 어떻게 촉진하는지 그 기전을 조사하였다. 특히 혈관평활근세포(VSMCs)에서 HNE가 사이클로옥시게나제-2(COX-2) 발현과 일산화질소(NO) 생성에 미치는 영향에 초점을 맞추었다.
VSMCs에 HNE를 처리한 후, 단백질 발현은 Western blot으로, mRNA 발현은 역전사 중합효소 연쇄반응(RT-PCR), ERK의 세포 내 위치는 세포 분획법, 신호 전달 경로는 특이적 억제제(U0126, SB203580, indomethacin)를 사용하여 분석하였다. 세포 내 NO 생성은 특이적 NO 지표제인 4,5-Diaminofluorescein(DAF-2) 형광 염색을 이용한 Flow Cytometry로 측정하였다.
HNE 처리는 VSMCs에서 COX-2 발현과 NO 생성을 유의미하게 증가시켰다. 이 효과는 HNE에 의해 유도된 ERK 인산화, 핵으로의 이동, 그리고 ERK 억제제 U0126에 의한 COX-2 및 NO 상향 조절의 완전한 억제를 통해 확인된 결과로 볼때 ERK 경로에 의해 매개되는 것으로 나타났다. p38 MAPK 억제제인 SB203580과 COX-2 억제제 indomethacin은 HNE에 의해 유도된 NO 생성을 억제하지 못했는데, 이는 ERK가 COX-2와 NO를 병렬적이고 독립적인 경로를 통해 활성화함을 나타낸다.
본 연구는 HNE가 ERK 신호전달 경로를 통해 혈관평활근세포(VSMCs) 내 COX-2 발현과 NO 생성을 유도함을 입증하였으며, 이는 상위 단계인 HNE-ERK 신호축을 표적으로 하는 것이 당뇨병성 혈관 합병증의 효과적인 치료전략이 될 수 있음을 시사한다.
Keywords:
Cyclooxygenase-2, 4-Hydroxynonenal, NO, Diabetic vascular dysfunction, ERK키워드:
사이클로옥시게나제-2, 4-하이드록시노네날, 산화질소, 당뇨병성 혈관이상, ERKIntroduction
Diabetic retinopathy (DR) remains a leading cause of acquired blindness among the working-age population worldwide. While DR is now widely recognized as a complex neurovascular disease, its pathology is fundamentally driven by progressive dysfunction of the retinal vasculature. This vascular dysfunction is the cornerstone of the early, non-proliferative stage of diabetic retinopathy, and its progression dictates the severity of the disease.[1] Specifically, the early stages are characterized by severe endothelial and vascular smooth muscle cell (VSMC) impairment, leading to three critical physiological abnormalities: impaired blood flow regulation, significantly increased vascular permeability (breakdown of the blood-retinal barrier), and eventual capillary dropout, culminating in localized retinal ischemia and subsequent vision loss.[2-4] Understanding the molecular triggers that initiate and sustain this early vascular dysfunction is crucial for therapeutic intervention.
Diabetic vascular complications, encompassing both macrovascular (e.g., atherosclerosis) and microvascular (e.g., retinopathy, nephropathy) diseases, represent the principal cause of morbidity and mortality in patients with diabetes.[5] A central component of this pathogenesis is oxidative stress, characterized by excessive generation of reactive oxygen species (ROS) in retinal tissue.[6] A critical consequence of this oxidative damage is lipid peroxidation, leading to the formation of biologically active aldehydes, with 4-hydroxynonenal (HNE) being one of the most abundant and potent.[7] Clinical evidence demonstrates significantly elevated HNE levels in diabetic patients with retinopathy,[8] underscoring its clinical relevance. However, despite the established role of HNE as the precise molecular mechanisms through which it contributes to diabetic vascular dysfunction remain incompletely characterized.
Concurrently, the inflammatory response plays a pivotal role in vascular dysfunction including diabetic retinopathy. Cyclooxygenase-2 (COX-2), the inducible isoform of the rate-limiting enzyme in prostaglandin synthesis, is a key mediator of inflammation. COX-2 expression is markedly upregulated in the retinas of diabetic patients and animal models.[9] The prostanoids derived from COX-2 activity, such as prostaglandin E2 (PGE2) and thromboxane A2, contribute to vascular dysfunction by promoting inflammation, enhancing vascular permeability, and potentiating vasoconstriction.[10,11]
While the individual roles of oxidative stress (via HNE) and COX-2-driven inflammation in vascular dysfunction are increasingly appreciated[12], the direct molecular link between HNE and COX-2 in the context of retinal VSMCs remains less explored. Notably, the ERK/MAPK signaling pathway, a key regulator of cell proliferation and inflammation, is a potential candidate for mediating this link.
Therefore, this study aims to investigate the up-regulation of COX-2 by HNE in VSMC, with a focused inquiry into the role of ERK signaling. We seek to elucidate whether HNE treatment directly induces COX-2 expression and activity in VSMCs and to determine the essential role of ERK activation. Understanding this specific signaling cascade will provide the molecular mechanisms driving vascular pathology and may identify new therapeutic targets for intervening in the destructive cycle of oxidative stress and inflammation.
Materials and Methods
1. Cell culture and HNE treatments
The rat aortic smooth muscle cell line A10 (American Type Culture Collection, USA) was cultured in Dulbecco's modified Eagle's medium (DMEM; Sigma-Aldrich, USA) supplemented with 10% fetal bovine serum (FBS), 100 U/mL penicillin, and 100 µg/mL streptomycin at 37°C in a humidified 5% CO2 atmosphere. For experiments, cells were seeded in 12-well plates at 4×104 cells/well in complete medium. After overnight attachment, cells were serum-starved in DMEM containing 0.5% FBS and pre-treated with specific inhibitors for 30 minutes, followed by treatment with 4-hydroxynonenal (HNE; >98% purity, Cayman Chemical, USA). HNE was dissolved in phosphate-buffered saline (PBS) immediately prior to each experiment, with final ethanol concentration maintained below 0.1%.
2. Assay of NO generation
Intracellular nitric oxide (NO) generation was measured using the NO-specific fluorescent probe 4,5-Diaminofluorescein (DAF-2)[13]. DAF-2 selectively reacts with NO to form the fluorescent triazolofluorescein, which is excited at 490-495 nm. A stock solution of DAF-2 (1 mg in 0.55 ml dimethyl sulfoxide) was stored at -20°C. For experiments, a working solution (0.5 µg/ml) was freshly prepared in 50 mM nitrogen-purged phosphate buffer (pH 7.4). Where indicated, cells were pre-incubated for 30 minutes with inhibitors: 50 µM PTIO (an NO scavenger), 10 µM U0126 (an ERK inhibitor), 1 µM indomethacin (a COX inhibitor), or 10 µM SB203580 (a p38 MAPK inhibitor). Cells grown in 12-well plates were then loaded with 10 µM of the cell-permeable diacetate form of the probe (DAF-2 DA) for 30 minutes at 37°C, followed by stimulation with HNE for an additional 30 minutes. Reactions were terminated by aspiration. Cells were trypsinized, collected on ice, and analyzed immediately by flow cytometry (BD FACSLyric, BD Biosciences, USA). Fluorescence intensity, which is proportional to the amount of NO trapped, was measured for 10,000 events per sample. Data are presented as the mean fluorescence intensity, analyzed from histogram plots of cell number versus FL-1 channel fluorescence.
3. Western blot analysis
Following treatment, cells were harvested, washed twice with ice-cold phosphate-buffered saline (PBS), and lysed in TNN lysis buffer (50 mM Tris/HCl pH 8.0, 120 mM NaCl, 0.5% Nonidet P-40) containing protease inhibitors (aprotinin, leupeptin, PMSF, pepstatin, DTT) and phosphatase inhibitors (NaF, Na3VO4) for 1 hour on ice with periodic vortexing every 10 minutes. After centrifugation at 8,000×g for 30 minutes to remove debris, protein concentration in the supernatant was quantified using the Lowry method with BSA standards. Equal protein amounts were resolved by 10-15% SDS-PAGE and transferred onto nitrocellulose membranes (BIO-RAD, Korea) for immunoblotting. Antibodies used for detecting proteins were mouse monoclonal anti COX-2 (610204, 1:500, BD Biosciences, San Jose, CA), rabbit anti-human PGI synthase (Santa Cruz SC-20933, 1:200).
4. Reverse Transcriptase-Polymerase Chain Reaction (RT-PCR)
Total RNA (2 µg) was reverse-transcribed using random primers. Briefly, RNA and primers were denatured at 75°C for 5 min and cooled on ice. Reverse transcription was performed in a 20 µL reaction containing 1×buffer, 0.5 mM dNTPs, 0.01 M DTT, 100 U reverse transcriptase, and 16.5 U RNase inhibitor at 37°C for 2 hours. Reactions were terminated at 100°C for 2 min. For PCR amplification, specific primers were used: COX-2 (F: 5'-TTCAAATGAGATTGTGGGAAAAT-3', R: 5'-AGATCATCTCTGCCTGAGTATCTT-3') and β-actin (F: 5'-AAGGAAGGCTGGAAGAGTGC-3', R: 5'-CTACAATGAGCTGCGTGTGG-3') as internal control. PCR products were resolved on 0.8% agarose gels and visualized with ethidium bromide under UV illumination.
5. Statistics
All data are presented as mean ± SEM of at least three independent experiments. Statistical significance was determined by one-way ANOVA followed by Dunnett's post-hoc test for multiple comparisons. A p-value less than 0.05 was considered statistically significant. (Sigma Stat 2.0, Jandel Scientific, Chicago, IL). Differences were considered statistically significant at p<0.05.
Results
1. Induction of COX-2 expression by 4-HNE in VSNCs
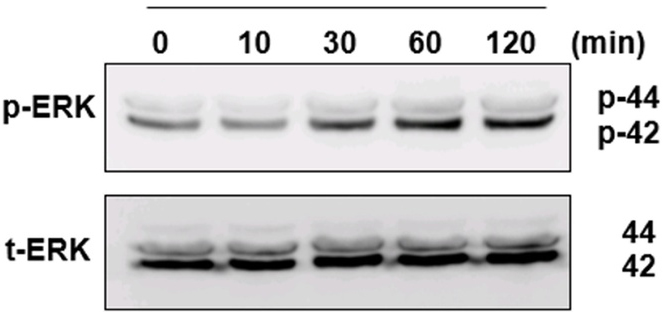
We first investigated whether 4-Hydroxynonenal (HNE) directly induces cyclooxygenase-2 (COX-2) expression in vascular smooth muscle cells (VSMCs). Western blot analysis revealed that treatment with HNE (5-30 µM) significantly increased COX-2 protein levels (Fig. 1). This finding demonstrates that HNE, a key lipid peroxidation product, acts as a potent inducer of COX-2 expression in VSMCs.
HNE induces COX-2 expression in VSMC.A. Representative western blots images showing COX-2 protein expression levels in VSMCs treated with the indicated concentrations of HNE for 24 h. Upregulation of COX-2 expression by HNE. B. Quantification of COX-2 expression was performed using densitometric analysis. The results represent means ± S.E.M. of three independent experiments. Statistical significance: *p<0.05, **p<0.01 vs. control.
2. HNE mediates ERK activation
To explore the mechanism underlying HNE-induced COX-2 expression, we examined the role of the extracellular signal-regulated kinase (ERK) signaling. Western blot analysis showed that HNE treatment significantly increased phosphorylated ERK (p-ERK) levels without altering total ERK expression (Fig. 2). This indicates that HNE activates the ERK signaling pathway in VSMCs. Although ERK phosphorylation in response to HNE has been previously reported,[14,15] our findings specifically verify this signaling event in VSMCs, showing that HNE treatment markedly enhances ERK phosphorylation.
3. Nuclear translocation of ERK by HNE treatment
Since ERK activation typically involves nuclear translocation to regulate gene transcription,[16] we investigated the subcellular localization of ERK following HNE treatment. Cellular fractionation assays demonstrated that HNE promoted the translocation of phosphorylated ERK from the cytosol to the nucleus (Fig. 3). This result confirms that HNE not only activates ERK but also facilitates its nuclear translocation, enabling ERK to modulate the transcription of target genes such as COX-2. These findings provide further mechanistic insight into how HNE drives inflammatory gene expression in VSMCs.

HNE promotes nuclear translocation of phosphorylated ERK. Cellular fractionation analysis showing the distribution of p-ERK in the cytosolic and nuclear fractions of VSMCs treated with HNE (20 µM) for the indicated durations. Cellular fractionation demonstrated that HNE treatment promotes the translocation of phosphorylated ERK from the cytosol to the nucleus. Nuclear accumulation of ERK was prominent at 60 min, coinciding with a reduction in its cytosolic fraction.
4. Transcriptional upregulation of COX-2 by HNE
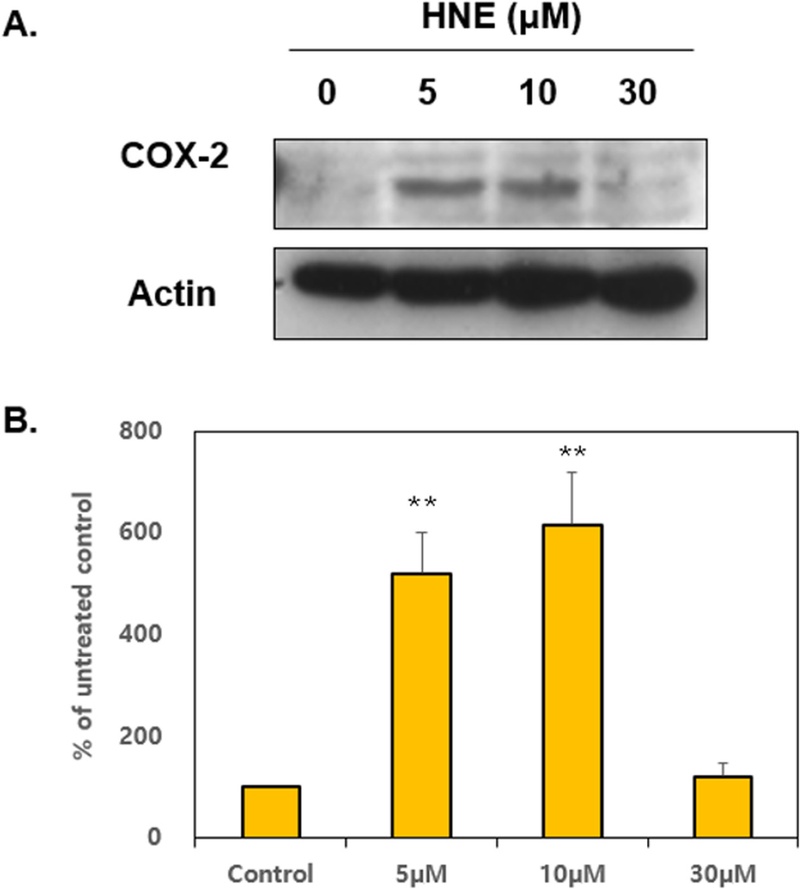
To determine whether HNE-induced COX-2 expression occurs at the transcriptional level, we measured COX-2 mRNA levels using RT-PCR. HNE treatment led to a significant increase in COX-2 mRNA expression (Fig. 4). The time-course experiment showed that COX-2 mRNA levels began to increase within 6 hours of HNE treatment, reaching maximum levels at 18 hours. This transcriptional activation, combined with the observed ERK nuclear translocation, supports the hypothesis that HNE activates the ERK pathway to enhance COX-2 transcription, thereby amplifying inflammatory signaling in VSMCs under vascular dysfunction.
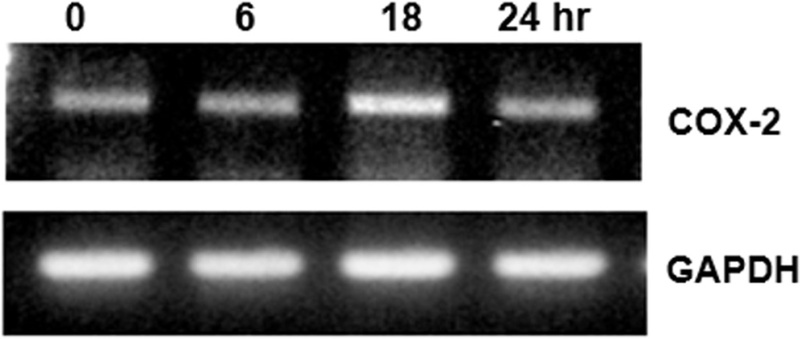
HNE upregulates COX-2 mRNA level. RT-PCR analysis of COX-2 mRNA levels in VSMCs treated with increasing concentrations of HNE for 6 h or with 20 µM HNE for various durations, showing that COX-2 mRNA increased from 6 h and peaked by 18 h. GAPDH was used as the positive control. The data are representative of at least 3 independent experiments.
5. Reduction of HNE-induced COX-2 mRNA by ERK inhibitor
To confirm the specific role of ERK signaling in HNE-induced COX-2 expression, we pre-treated VSMCs with the ERK inhibitor U0126 and the p38 MAPK inhibitor SB203580.[17] Pre-treatment with U0126 significantly suppressed HNE-induced COX-2 mRNA expression, whereas SB203580 had no inhibitory effect (Fig. 5). As a positive control, the NO donor SNAP also increased COX-2 mRNA levels, and this induction was similarly increased by HNE.
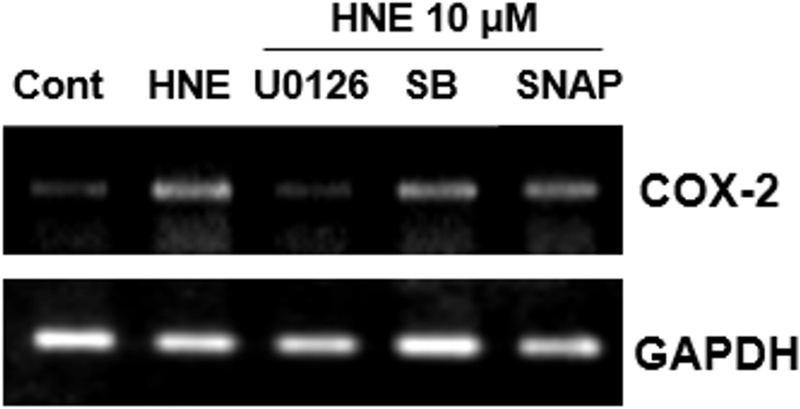
HNE upregulates COX-2 mRNA level. RT-PCR analysis of COX-2 mRNA levels in VSMCs treated with increasing concentrations of HNE for 6 h or with 20 µM HNE for various durations, showing that COX-2 mRNA increased from 6 h and peaked by 18 h. GAPDH was used as the positive control. The data are representative of at least 3 independent experiments.
6. Reduction of HNE-induced NO generation by ERK inhibitor
Based on our previous findings demonstrating that HNE promotes nitric oxide (NO) generation and considering the well-established role of NO in vascular dysfunction,[18,19] we investigated whether the ERK signaling pathway mediates HNE-induced NO production in VSMCs. To test this hypothesis, we measured NO levels in VSMCs following HNE treatment with or without pre-treatment with specific inhibitors. HNE-induced NO production was suppressed by the ERK inhibitor U0126 but was unaffected by the COX-2 inhibitor indomethacin. [20] These results demonstrate that HNE-induced NO production is mediated through the ERK pathway (inhibited by U0126) but operates independently of COX-2 activity (not inhibited by indomethacin) (Fig. 6).
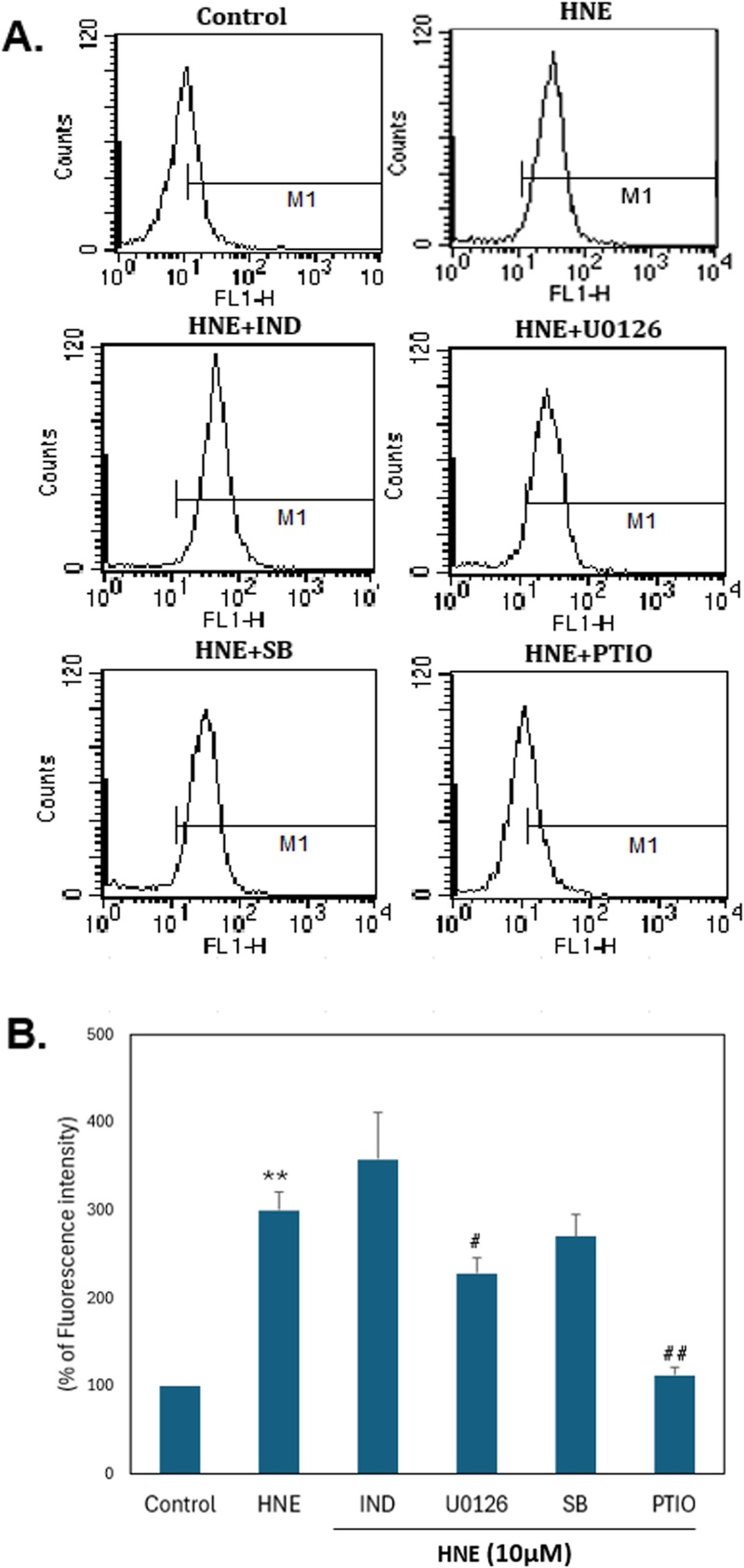
ERK iinhibitor attenuates HNE-induced NO production through a COX-2 independent pathway. NO production in VSMCs pre-treated with U0126 (10 µM), SB203580 (10 µM), or indomethacin (10 µM, COX-2 inhibitor) for 30 min, followed by HNE (20 µM) treatment for 1 h with DAF-2, and analyzed using FACS. 2-(4-Carboxyphenyl)-4,4,5,5-tetramethylimidazoline-1-oxyl 3-oxide (PTIO, NO scavenger), 50 µM, was used as the negative control. HNE-induced NO production was suppressed by the ERK inhibitor U0126 but was unaffected by the COX-2 inhibitor indomethacin. A. Representative histogram plots of cell numbers versus fluorescence intensity (FL-1). B. The data represent the percentage of DAF-2 fluorescence intensity relative to that of the control. Data are presented as mean±S.E.M. (n=3). *p<0.05, **p<0.01 vs. control; #p<0.05, ##p<0.01 vs. HNE-only group.
Discussion
Our findings demonstrate that 4-hydroxynonenal (HNE) is a potent inducer of both cyclooxygenase-2 (COX-2) expression and nitric oxide (NO) production in vascular smooth muscle cells (VSMCs). The dose-dependent upregulation of COX-2 protein and mRNA establishes a direct molecular link between this key lipid peroxidation product and a major inflammatory pathway.[12] HNE-induced COX-2 upregulation is specifically mediated through the ERK pathway (Fig. 5). While ERK inhibition suppressed both COX-2 expression and NO production, the COX-2 inhibitor indomethacin failed to inhibit HNE-induced NO generation. These results confirm the central role of ERK activation in linking oxidative stress (via HNE) to inflammatory responses in VSMCs.
Taken together, our data indicates that HNE activates COX-2 and iNOS/NO through independent pathways downstream of ERK, rather than in a sequential cascade. This parallel activation mechanism enables HNE to amplify the inflammatory response by simultaneously engaging multiple effector pathways. In line with previous reports,[9,15] our results demonstrate that VSMCs exposed to elevated HNE levels in the diabetic vasculature are driven into a pro-inflammatory state, characterized by heightened COX-2 expression.
Conclusions
This research demonstrates that the lipid peroxidation product HNE activates the ERK signaling pathway in vascular smooth muscle cells. This ERK activation simultaneously increases both COX-2 inflammatory enzyme and nitric oxide (NO) production. Experiments using ERK inhibitors confirmed the importance of this pathway, while also revealing that COX-2 and NO effect through distinct mechanisms. These findings suggest that the HNE-ERK signaling pathway could be an important therapeutic target for vascular dysfunction.
Acknowledgments
This research was supported by the Regional Innovation System & Education (RISE) program of Suseong University in 2025.
References
-
Zeng Y, Cao D, Yang D, et al. Retinal vasculature-function correlation in non-proliferative diabetic retinopathy. Doc Ophthalmol. 2020;140(2):129-138.
[https://doi.org/10.1007/s10633-019-09724-4]
-
Faries PL, Rohan DI, Takahara H, et al. Human vascular smooth muscle cells of diabetic origin exhibit increased proliferation, adhesion, and migration. J Vasc Surg. 2001;33(3):601-607.
[https://doi.org/10.1067/mva.2001.111806]
-
Gilbert RE. The endothelium in diabetic nephropathy. Curr Atheroscler Rep. 2014;16(5):410.
[https://doi.org/10.1007/s11883-014-0410-8]
-
Antonetti DA, Klein R, Gardner TW. Diabetic retinopathy. N Engl J Med. 2012;366(13):1227-1239.
[https://doi.org/10.1056/NEJMra1005073]
-
Cooper ME, Gilbert RE, Jerums G. Diabetic vascular complications. Clin Exp Pharmacol Physiol. 1997;24(9-10):770-775.
[https://doi.org/10.1111/j.1440-1681.1997.tb02130.x]
-
Poli G , Schaur RJ , Siems WG , et al. 4-hydroxynonenal: a membrane lipid oxidation product of medicinal interest. Med Res Rev. 2008;28(4):569-631.
[https://doi.org/10.1002/med.20117]
-
Uchida K. 4-Hydroxy-2-nonenal: a product and mediator of oxidative stress. Prog Lipid Res. 2003;42(4):318-343.
[https://doi.org/10.1016/s0163-7827(03)00014-6]
-
Augustine J, Troendle EP, Barabas P, et al. The role of lipoxidation in the pathogenesis of diabetic retinopathy. Front Endocrinol. 2021;11:621938.
[https://doi.org/10.3389/fendo.2020.621938]
-
Ayalasomayajula SP, Kompella UB. Celecoxib, a selective cyclooxygenase-2 inhibitor, inhibits retinal vascular endothelial growth factor expression and vascular leakage in a streptozotocin-induced diabetic rat model. Eur J Pharmacol. 2003;458(3):283-289.
[https://doi.org/10.1016/s0014-2999(02)02793-0]
-
Ricciotti E, FitzGerald GA. Prostaglandins and inflammation. Arterioscler Thromb Vasc Biol. 2011;31(5):986-1000.
[https://doi.org/10.1111/ATVBAHA.110.207449]
-
Hoxha M, Buccellati C, Capra V, et al. In vitro pharmacological evaluation of multitarget agents for thromboxane prostanoid receptor antagonism and COX-2 inhibition. Pharmacol Res. 2016;103:132-143.
[https://doi.org/10.1016/j.phrs.2015.11.012]
-
Uchida K. HNE as an inducer of COX-2. Free Radic Biol Med. 2017;111:169-172.
[https://doi.org/10.1016/j.freeradbiomed.2017.02.004]
-
Sutherland H, Khundkar R, Zolle O, et al. A fluorescence-based method for measuring nitric oxide in extracts of skeletal muscle. Nitric Oxide. 2001;5(5):475-481.
[https://doi.org/10.1006/niox.2001.0374]
-
Sun Z, Jiang W, Lu G, et al. Loss of ALDH2 accelerates the progression of pulmonary arterial hypertension through the 4-HNE/ERK1/2-p16(INK4a) signaling pathway. Biochim Biophys Acta Mol Basis Dis. 2025;1871(6):167863.
[https://doi.org/10.1016/j.bbadis.2025.167863]
-
Singh DK, Winocour P, Farrington K. Oxidative stress in early diabetic nephropathy: fueling the fire. Nat Rev Endocrinol. 2011;7(3):176-184.
[https://doi.org/10.1038/nrendo.2010.212]
-
Honma S, Kakuage S, Morita Y, et al. Vildagliptin treatment ameliorates renal interstitial fibrosis in a murine model of unilateral ureteral obstruction. Biol Pharm Bull. 2024;47(1):37-42.
[https://doi.org/10.1248/bpb.b23-00609]
-
Lee SJ, Kim CE, Yun MR, et al. 4-Hydroxynonenal enhances MMP-9 production in murine macrophages via 5-lipoxygenase-mediated activation of ERK and p38 MAPK. Toxicol Appl Pharmacol. 2010;242(2):191-198.
[https://doi.org/10.1016/j.taap.2009.10.007]
-
Chen RH , Sarnecki C, Blenis J. Nuclear localization and regulation of erk- and rsk-encoded protein kinases. Mol Cell Biol. 1992;12(3):915-927.
[https://doi.org/10.1128/mcb.12.3.915-927.1992]
-
Lee J. Induction of 5-lipoxygenase by 4-hydroxynonenal via nitric oxide generation in vascular smooth muscle cells. J Korean Ophthalmic Opt Soc. 2023;28(3):255-262.
[https://doi.org/10.14479/jkoos.2023.28.3.255]
-
Sun L, Chen K, Jiang Z, et al. Indometacin inhibits the proliferation and activation of human pancreatic stellate cells through the downregulation of COX-2. Oncol Rep. 2018;39(5):2243-2251.
[https://doi.org/10.3892/or.2018.6321]